
Imagine this: you’ve just finished crafting a stunning redwood picnic table. Weeks of sawing, sanding, and staining have paid off.
Now, as winter approaches, a question lingers: how do you protect this masterpiece from the brutal elements?
You’ve heard whispers, rumors in the woodworking world – stories of old-timers using a surprising substance to keep their wood strong: antifreeze.
Antifreeze, or engine coolant, is that brightly colored liquid we routinely pour into our car’s radiator to prevent it from freezing in winter and overheating in summer.
It’s a sophisticated cocktail designed for high-performance engines, but does it possess secret powers as a good wood preservative?
Is this a genius hack, or a dangerous DIY disaster waiting to happen? Let’s break down the chemistry.
Antifreeze as a Wood Preservative
At its core, antifreeze is typically composed of either ethylene glycol or propylene glycol, mixed with water and a package of additives (rust inhibitors, anti-foaming agents, etc.).
These glycols are alcohols, characterized by their high boiling points and incredibly low freezing points – hence their primary function in vehicles.
But can they translate these properties to the world of woodworking?
The theory behind using antifreeze as a wood preservative stems from its ability to displace water.
Wood is a hygroscopic material, meaning it readily absorbs and releases moisture from its environment. This fluctuation in moisture content is a primary driver of decay, wood rot, and the dreaded dimensional instability (more on that later).
Glycols, being miscible with water, can penetrate the porous structure of wood and replace some of the water molecules bound within its cell walls.
By doing so, they can theoretically reduce the wood’s equilibrium moisture content (EMC), making it less susceptible to the growth of fungi and bacteria that thrive in damp conditions.
However, before you start basting your furniture with the green stuff, let’s look at the cold, hard facts…

Is Antifreeze a Viable Wood Preservative?
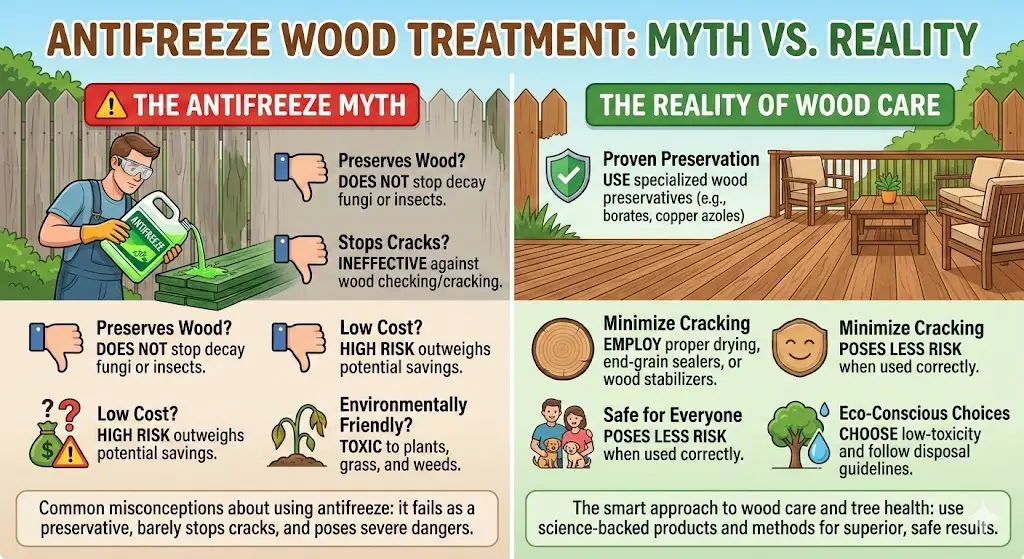
The overwhelming consensus from wood scientists and professional contractors is a resounding “no”.
The risks – from extreme toxicity and environmental harm to increased fire hazard and questionable effectiveness – far outweigh any potential benefits.
True wood preservatives are formulated with specific chemical compounds like borates, copper azoles, or creosote (though creosote is highly regulated) that are proven to combat rot-causing fungi and insects.
They are designed for this purpose, with safety data sheets and clear application guidelines. Antifreeze is a vehicle maintenance product, not a structural protectant.
Pros of using antifreeze on wood
1- Low Freezing Point: Can theoretically reduce the chance of freeze-thaw damage in very cold climates.
2- Water Displacement: Can lower the equilibrium moisture content (EMC) of wood, potentially inhibiting fungal growth.
3- Availability: Readily available and inexpensive compared to some specialized wood preservatives.
4- Historical Anecdotes: Some woodworkers have reported using it with success, contributing to its underground reputation.
Cons of antifreeze as a wood preservative
1- High Toxicity: Ethylene glycol, the most common type, is extremely toxic to humans, pets, and wildlife. Ingesting even a small amount can be fatal.
2- Environmental Hazard: Leaching can contaminate soil and water, posing a serious threat to ecosystems. It can also act as a killer of unintended plants, grass, and weeds.
3- Fire Hazard: Glycols are flammable, and their presence can significantly increase the fire risk of the treated wood.
4- Not an Effective Fungicide/Insecticide: Antifreeze is not designed to kill the specific fungi and insects that cause wood rot and damage. It lacks the target-specific biocides found in true wood preservatives.
5- Difficulty in Application and Penetration: Ensuring deep and even penetration into the wood structure can be challenging without proper pressure treatment.
6- Can Affect Finishes: Antifreeze can interfere with the adhesion and performance of subsequent finishes like paint or varnish.
7- Unknown Long-Term Effects: The long-term durability and performance of wood treated with antifreeze are largely unstudied and unpredictable.
8- Ineffective Against Termites: Offers no protection against wood-boring insects like beetles and termites.
Will Antifreeze Prevent the Wood Cracks?
Now, let’s address the specific question that might have sparked this entire curiosity: can antifreeze prevent those unsightly cracks that develop in wood, especially as it dries?
The short answer is: not really, and certainly not effectively.
Wood cracks, or checks, are primarily caused by the uneven shrinkage that occurs as wood dries, particularly across its grain.
This process is most pronounced in green wood – wood that has recently been cut and still holds a high percentage of its internal moisture.
As the wood is exposed to drier air, the surface layers dry more quickly than the core. This creates tension, and when the tension exceeds the wood’s strength, it tears apart, resulting in a crack.
The “antifreeze for cracks” idea might stem from the belief that the glycols can help stabilize the wood and slow down the drying process.
While it’s true that glycols can alter the moisture-holding properties of wood, they are not an effective stabilizer on their own.
The penetration is often superficial, and the effect is not uniform enough to counteract the significant and uneven forces of shrinkage that lead to cracking.

Can I Use Antifreeze on Trees – Will it Kill My Trees?
This is a scenario where the consequences are not just structural or cosmetic – they are potentially lethal.
Using antifreeze around trees, whether intended as a treatment or due to accidental spills, is extremely dangerous.
Yes, antifreeze is toxic to trees and will likely kill them.
The active ingredients in most antifreeze, ethylene glycol and propylene glycol, are highly toxic to all forms of life, including plants.
If antifreeze is applied to the soil around a tree, it can leach into the groundwater and be absorbed by the tree’s roots.
From there, it moves through the tree’s vascular system, disrupting critical biological processes.
Specifically, glycols can:
- Inhibit Photosynthesis: They interfere with the tree’s ability to produce energy.
- Cause Cellular Damage: They can disrupt cell membranes and lead to tissue death.
- Affect Nutrient Uptake: They can impair the roots’ ability to absorb essential nutrients and water.
The symptoms of antifreeze poisoning in trees can vary, but often include:
- Chlorosis: Yellowing of leaves, indicating a lack of chlorophyll.
- Leaf Drop: Premature loss of leaves.
- Stunting: Slowed or stopped growth.
- Wilting: A classic sign of water stress, even with adequate soil moisture.
- Branch Dieback: Sections of the tree are becoming brittle and dying.
- Bark Splitting and Cankers: Indicators of internal tissue damage.
Could I dilute the antifreeze with water to make it safer?
While dilution would technically lower the concentration of the glycols, it doesn’t eliminate the inherent danger. Antifreeze is still an extreme health and environmental hazard even in diluted forms.
Dilution also significantly reduces its potential (already minimal) effectiveness as a wood treatment. It’s much better to avoid using it altogether and opt for safer, proven alternatives.
Are there any wood species that might be particularly sensitive to antifreeze?
Because the toxic effects of glycols are universal to plant life, it’s safe to say that all tree species would be negatively impacted.
However, younger, smaller trees with less established root systems would be most vulnerable, as a smaller amount of the toxin could cause proportionally more damage.
Fruit trees or any trees destined for consumption would be a particularly bad idea to use antifreeze near, as the glycols could potentially contaminate the fruit.
What should I do if I accidentally spill antifreeze on my lawn or a wooden structure?
If you suspect a tree has been exposed to antifreeze, it is a race against time. The best course of action is to:
a) Dilute the Contamination: Immediately drench the affected soil area with large amounts of water to try to leach the antifreeze away from the tree’s root zone.
b) Apply Activated Charcoal: In some cases, applying activated charcoal to the soil can help absorb some of the toxin before it’s taken up by the roots.
c) Consult an Arborist: A professional arborist can assess the damage, determine the severity of the exposure, and recommend appropriate treatment options, which may include further watering, aeration, or even specific chemical treatments in severe cases.
However, be aware that in many instances, the damage from antifreeze is severe, and the tree may not be salvageable.
This warning extends not just to trees but to all plant life. Antifreeze is non-selective. It won’t distinguish between your prize-winning rose bush and a stubborn weed.
In fact, it can act as a potent killer of unintended plants, grass, and weeds, leaving a path of ecological destruction in its wake. This characteristic alone makes it a terrible choice for any application near vegetation.
Final Thoughts
The desire to protect our creations and property is a natural one. The woodworking and landscaping worlds are full of challenges, and it’s tempting to look for creative solutions.
However, creativity must always be tempered with safety and scientific understanding. Using antifreeze as a wood preservative or around trees is a classic example where a seemingly plausible idea crumbles under the weight of toxic reality.
The core lesson here is to always respect the intended purpose of chemical products. A product formulated for a complex engine system is not a cure-all for all structural or biological woes.
True wood preservation is achieved with biocides designed to fight decay fungi and insects. Minimizing cracks is accomplished with controlled drying and specialized stabilizers. And tree health is safeguarded by ensuring a toxic-free environment.
For the sake of your health, your property, and the planet, stick to the products and practices that are proven to be both effective and safe.
Hi, I am Mark Garner a professional carpenter, woodworker, and DIY painter. I live in the small city of Peoria, Arizona as a semi-retired woodworker. I have started this blog with a simple motive to help you with my wood experience in this sector. If you like to know more about what I love doing and how it all got started, you can check more about me here.




